 The metal filament heats up and its electrons escape in the form of thermionic emission. 
Now can you see a spot?Ī spot is only visible on the fluorescent screen when the cathode is connected. Reconnect the voltage lead to the cathode and repeat the previous step.Can you see a spot on the fluorescent screen? Connect the power unit to a source of electricity.Set the voltage of the anode to 30–50 V.Set the voltage of the auxiliary anode – the anode of the control grid or Wehnelt cylinder – to 10 V.On the CRT power supply unit, disconnect the lead that supplies the voltage to the cathode (see the circuit diagram in the attached worksheet).See the list of the necessary materials in the downloadable document w1 Procedure For all activities, the particle accelerator needs to be set up as outlined in the worksheet that can be downloaded w1. All four activities could occupy a class for at least half a day, but they could also be used separately in individual lessons. The activities described below enable students to control the same parameters in a CRT as scientists do at the LHC: creating a particle beam, changing the path of the particles and altering their speed. To collide the beam of particles with a second beam and observe the result To cause a beam of particles to form an image on a fluorescent screen Strong magnetic fields achieved using superconducting electromagnets (4 T in strength) Protons produced by ionisation of hydrogen atomsĪ potential difference between the anode and cathodeĮlectronic fields and radio frequencies, synchronised with particle speed.Pressure (For comparison, a vacuum cleaner has a pressure of 1‒10 -3 atm, and outer space has a pressure of <10 -15atm)ĭistance travelled by a particle between collisionsĮlectrons produced by thermionic emission at the cathode (a heated filament) Photo courtesy of CERN Table 1: A comparison of the classroom particle accelerator (the CRT) and CERN’s LHC Characteristic The image could be, for example, electrical waveforms (on an oscilloscope), radio wave echoes of aircraft or ships (on a radar screen) or pictures on an old-fashioned television screen or computer monitor 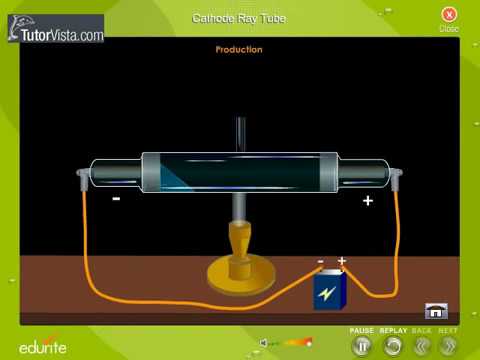
The electrons can then be deflected by a magnetic (or in the case of oscilloscopes, an electrical) field (D) before they strike the phosphorescent screen (E), creating an image. If we alter the potential difference between the Y-plates, the beam is deflected upwards or downwards on the screen.Figure 1: The cathode ray tube is a vacuum tube in which electrons are produced by a heated filament (the cathode, A), focused into a beam as they pass through the aperture of the control grid (Wehnelt cylinder, B) and accelerated by the voltage (VA) between the cathode and the anode (C). Usually, the potential difference applied to the X-plates makes the spot move across the screen at a uniform speed. This deflection is produced by two pairs of parallel plates arranged at right angles. If necessary the stream of emerging electrons can be deflected in its passage between the gun and the screen. On leaving the gun, the electron stream passes across the tube and eventually hits the screen at the far side. Such arrangement of electrodes where a stream of electrons is produced is often known as the electron gun. As a result, the electrons accelerate across the gap between the electrodes and a narrow stream of the electrons emerges from the hole in the anode. A potential difference of some hundreds of volts is applied between cathode and anode. At a short distance from the cathode is an anode having a central hole in it. In the oscilloscope, the electrons are emitted by a hot cathode which is situated in a highly evacuated tube. The oscilloscope has many points in common with the discharge tube. The glow produced by the fast moving electrons on a fluorescent screen led to its use in radar and television. The discovery of cathode rays led to a vast field of practical application in Electronics. Modern cathode ray tubes have hot cathodes which require much less voltage, nearly 3000 volts. It needed a high voltage of the order of 30,000 volts. The cathode, in the discharge tubes used by Sir Thomson, was cold cathode. Experiments showed that mass of the electron is approximately 1/1860 of mass of hydrogen atom. Thomson found that the electrons had mass far less than of even the hydrogen atom. 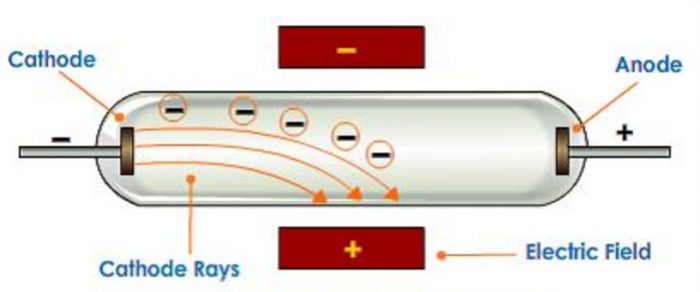
We know that hydrogen atom is the lightest atom. As these emanate from the cathode, the rays are called the Cathode Rays. These electrons emanate normally from the cathode. When this fluorescence was investigated, it was found that the fluorescence consisted of beams of negatively charged electrons. 
When the pressure in the discharge tube is less than 10 -4 mm of Hg, the discharge tube starts showing fluorescence. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
